Mean Square Fluctuations of Energy (monoatomic gas)
Monoatomic Perfect Gas (N particles)
Below are (A) the mean-square fluctuation in energy in the canonical ensemble and (B) the probability that a chosen particle’s velocity component ![]() lies in the interval
lies in the interval ![]() . Equations are provided in LaTeX and will be rendered by MathJax.
. Equations are provided in LaTeX and will be rendered by MathJax.
A. Mean-square fluctuation of the energy (canonical ensemble)
Start with the canonical partition function ![]() for the full system at temperature
for the full system at temperature ![]() . Define
. Define ![]() . The canonical relations are
. The canonical relations are
![]()
![]()
Using the thermodynamic identity ![]() (or equivalently using standard manipulations) one obtains the well-known relation
(or equivalently using standard manipulations) one obtains the well-known relation
![]()
where ![]() is the heat capacity at constant volume.
is the heat capacity at constant volume.
For a classical monoatomic ideal gas
![]()
![]()
Relative fluctuation (useful scaling):
![]()
This shows energy fluctuations scale as ![]() and are negligible for macroscopic
and are negligible for macroscopic ![]() .
.
B. Probability that a particle’s 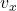 lies in
lies in ![Rendered by QuickLaTeX.com [v_x,\,v_x+\Delta v_x]](https://stationarystates.com/wp-content/ql-cache/quicklatex.com-c3f813fcdaab429fc9292adc64441b42_l3.png)
In the canonical ensemble for a classical ideal gas the single-particle momentum/velocity components are independent and Gaussian. The one-component Maxwell–Boltzmann probability density for ![]() is
is
![]()
For a small interval ![]() (infinitesimal approximation), the probability that a chosen particle has
(infinitesimal approximation), the probability that a chosen particle has ![]() in
in ![]() is
is
![Rendered by QuickLaTeX.com \[ \boxed{\qquad P\big(v_x\le v_x' < v_x+\Delta v_x\big) \;\approx\; f_{v_x}(v_x)\,\Delta v_x \;=\; \sqrt{\frac{m}{2\pi k_B T}}\,e^{-\frac{m v_x^2}{2k_B T}}\;\Delta v_x \qquad} \]](https://stationarystates.com/wp-content/ql-cache/quicklatex.com-7f10a8d45ecdd70768e057cbe1e6e12a_l3.png)
For the 3D speed (magnitude) distribution the Maxwell speed density is
![]()
so the probability a chosen particle’s speed lies in ![]() is
is ![]() .
.
C. Probability that any of the N particles has 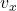 in the interval
in the interval
Let
![]()
be the single-particle probability for that small interval. Assuming independent particles, the probability that none of the ![]() particles lies in the interval is
particles lies in the interval is ![]() . Thus the probability that at least one particle lies in the interval is
. Thus the probability that at least one particle lies in the interval is
![]()
If ![]() is very small and
is very small and ![]() then
then ![]() (expected number of particles in the interval).
(expected number of particles in the interval).
D. Optional: brief canonical derivation of 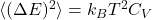
From the partition function ![]() :
:
![]()
Hence
![]()
Noting ![]() and recognizing
and recognizing ![]() yields the relation
yields the relation ![]() .
.
E. Quick summary
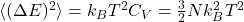 for a monoatomic ideal gas.
for a monoatomic ideal gas.- Single-component velocity density:
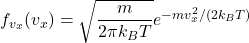 .
. - Probability (small interval):
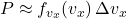 . For any of the N particles:
. For any of the N particles: 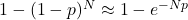 .
.
If you’d like, I can also:
- Provide the same page but with explicit numeric examples (choose
 ).
). - Show the derivation of the Maxwell distribution from the canonical single-particle Hamiltonian step-by-step.
- Format this for printing (PDF-friendly) or convert to LaTeX source.