Angular momentum for particle in a box
https://stationarystates.com/basic-quantum-theory/angular-momentum…article-in-a-box/
Energy Levels of a Particle in a Box with Angular Momentum
1. Particle in a 1D Box
In 1D, angular momentum doesn’t exist in the usual sense because rotation requires at least two dimensions.
Energy levels remain:
![]()
2. Particle in a 2D or 3D Box
For a 2D rectangle or 3D cube, the Schrödinger equation separates in Cartesian coordinates:
![]()
Angular momentum is not conserved in a cubical box. Energy depends on quantum numbers along each axis:
![Rendered by QuickLaTeX.com \[ E_{n_x,n_y,n_z} = \frac{\hbar^2 \pi^2}{2 m} \left( \frac{n_x^2}{L_x^2} + \frac{n_y^2}{L_y^2} + \frac{n_z^2}{L_z^2} \right) \]](https://stationarystates.com/wp-content/ql-cache/quicklatex.com-4c961439ce6609a6cf0bd7b76e4c58b6_l3.png)
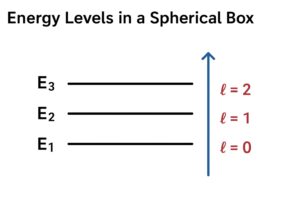
3. Particle in a Spherical Box
If the box is spherically symmetric, angular momentum L is a good quantum number.
The Schrödinger equation in spherical coordinates:
![]()
Separate variables:
![]()
where Y_\ell^m are spherical harmonics and \ell is the angular momentum quantum number.
Energy levels include a centrifugal term:
![]()
Here, ![]() are the zeros of spherical Bessel functions.
are the zeros of spherical Bessel functions.
Larger angular momentum (![]() ) increases energy because the wavefunction is “pushed outward”.
) increases energy because the wavefunction is “pushed outward”.
4. Key Takeaways
- 1D box: Angular momentum is irrelevant; energy levels are unchanged.
- Rectangular/cubical box: Energy depends on quantum numbers along each axis, not angular momentum.
- Spherical box: Higher angular momentum quantum number
\ellraises the energy.
Intuition: Higher angular momentum → particle “rotates” more → less probability near the center → higher energy.