Particle in a Box? Momentum and Distance Traveled – 1-Inch box to Atomic Scale Box
How “Stationary” Is a Particle in a Box? Momentum and Travel Distance from 1-Inch to Atomic Scales
In the 1D infinite square well, the ground state is a standing wave. That means the expectation value of momentum is zero, yet the state can be written as an equal superposition of two traveling waves with definite momenta ![]() . The magnitude
. The magnitude ![]() is well defined by the box width
is well defined by the box width ![]() .
.
1) Ground-State Momentum in a 1-Inch-Wide Box
Box width:
![]()
For the ![]() -th stationary state of a 1D infinite well, the magnitude of the momentum is
-th stationary state of a 1D infinite well, the magnitude of the momentum is
![]()
Using ![]() and
and ![]() :
:
![]()
| Quantity | Value |
|---|---|
| Width |
|
| Ground-state momentum magnitude |
2) “Distance Traveled” Inside the 1-Inch Box
Because the ground state is a standing wave, the probability density is static. But if you decompose it into its two traveling components, each one behaves like a particle with momentum magnitude ![]() and speed
and speed ![]() . The distance covered in time
. The distance covered in time ![]() would be
would be ![]() .
.
Electron
![]()
![]()
Proton
![]()
| Particle | Mass (kg) | Speed |
Time to cross |
|---|---|---|---|
| Electron | |||
| Proton |
Takeaway: With a macroscopic well, the quantized momentum is tiny. Even an electron moves only about a centimeter per second in this ground-state momentum scale.
3) Atomic-Scale Box: 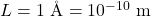
Now the same formulas, but with a microscopic width.
Ground-state momentum
![]()
Electron
![]()
![]()
Proton
![]()
![]()
| Particle | Speed (m/s) | Traverse time for |
Kinetic energy (eV) | |
|---|---|---|---|---|
| Electron | ||||
| Proton |
4) Why the Huge Difference?
- The momentum scale is set by
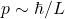 . Shrinking
. Shrinking 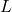 by a factor of
by a factor of 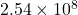 (from 1 inch to 1 Å) increases
(from 1 inch to 1 Å) increases 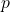 by the same factor.
by the same factor. - Electron speeds at atomic scales naturally land in the
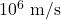 range, and characteristic times are attoseconds to femtoseconds—matching intuition from atomic physics.
range, and characteristic times are attoseconds to femtoseconds—matching intuition from atomic physics. - At macroscopic
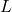 , quantization steps are so fine that the ground-state momentum (and kinetic energy) are minuscule.
, quantization steps are so fine that the ground-state momentum (and kinetic energy) are minuscule.
5) The Standing-Wave Subtlety
The ground state wavefunction in the infinite well is
![]()
which is a standing wave with ![]() . Yet it decomposes into traveling waves carrying momenta
. Yet it decomposes into traveling waves carrying momenta ![]() . So while there is no unique classical trajectory for the stationary state, the momentum magnitude scale that governs any localized wavepacket built from the ground state is precisely
. So while there is no unique classical trajectory for the stationary state, the momentum magnitude scale that governs any localized wavepacket built from the ground state is precisely
![]()
Bottom Line
- 1-inch box:
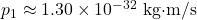 . An electron’s speed would be
. An electron’s speed would be 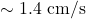 , taking
, taking 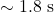 to cross once.
to cross once. - 1 Å box:
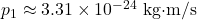 . An electron’s speed is
. An electron’s speed is 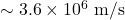 , crossing in
, crossing in 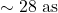 with kinetic energy
with kinetic energy 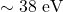 .
.
Same quantum rule, wildly different scales—because everything scales as ![]() .
.