Estimate the nuclear radius size based on Electrostatic and Magnetic Field Strengths
The Rasetti–Fermi Paradox (1926–1927): Two “Radius” Estimates That Don’t Match
A historical back-of-the-envelope puzzle: estimating a nuclear “radius” from an electron’s electrostatic binding vs. its magnetic interaction with the nuclear dipole leads to incompatible scales—evidence against the old “nuclear electron” hypothesis.
Overview
In the late 1920s, Rasetti and Enrico Fermi noted a striking inconsistency. If one assumes an electron could be bound inside the nucleus, then:
- Equating the Coulomb (electrostatic) energy to the electron rest energy yields a characteristic length
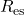 of a few femtometers.
of a few femtometers. - Equating the magnetic dipole interaction energy to the electron rest energy yields a (different) characteristic length
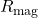 that is larger by a factor
that is larger by a factor 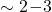 .
.
Both scales were supposed to refer to the same physical nuclear size if an electron truly lived inside the nucleus. Their mismatch posed a paradox—one of several reasons the community eventually abandoned the idea of pre-existing nuclear electrons, well before the neutron’s discovery and Fermi’s β-decay theory resolved the issue.
Electrostatic (“Coulomb”) Estimate
Treat the electron (charge ![]() ) as confined within a spherical nucleus of radius
) as confined within a spherical nucleus of radius ![]() and total charge
and total charge ![]() . A crude binding scale is obtained by matching the Coulomb energy at the nuclear surface to the electron rest energy:
. A crude binding scale is obtained by matching the Coulomb energy at the nuclear surface to the electron rest energy:
![]()
For ![]() (hydrogenic charge), this is the classical electron radius:
(hydrogenic charge), this is the classical electron radius:
![]()
Magnetic Dipole Estimate
Model the nucleus as a magnetic dipole of moment ![]() (order of the nuclear magneton
(order of the nuclear magneton ![]() , times a factor of order unity). The on-axis dipole field is
, times a factor of order unity). The on-axis dipole field is
![]()
A bound electron with magnetic moment ![]() has Zeeman energy
has Zeeman energy ![]() . Setting this equal to
. Setting this equal to ![]() gives
gives
![]()
For a protonic moment (![]() ), this yields a scale
), this yields a scale ![]() (order of magnitude).
(order of magnitude).
The Paradox (Rasetti–Fermi)
| Method | Condition | Implied Scale | Typical Value |
|---|---|---|---|
| Electrostatic | |||
| Magnetic dipole |
Paradox: If an electron were truly confined inside the nucleus, both estimates should describe the same length scale. Instead, ![]() comes out significantly larger than
comes out significantly larger than ![]() . This robust mismatch (insensitive to order-unity factors) undermined the hypothesis of nuclear electrons.
. This robust mismatch (insensitive to order-unity factors) undermined the hypothesis of nuclear electrons.
How Bethe & Bacher (1936) Restated It
In their classic 1936 review (Reviews of Modern Physics 8, 82), Bethe and Bacher summarized the situation (paraphrased):
Assuming electrons reside in the nucleus leads to contradictory size estimates. From electrostatics one finds a confinement scale comparable to the classical electron radius, while the magnetic interaction with the nuclear dipole moment implies a larger characteristic scale. The two cannot be reconciled within a single nuclear radius, indicating that the assumption of pre-existing nuclear electrons is untenable.
(Paraphrase for clarity; the review collects multiple arguments—spin/statistics, β spectra, and magnetic/electrostatic considerations—against nuclear electrons, anticipating the post-1932 neutron picture and Fermi’s β-decay theory.)
Numerical Illustration (Hydrogenic Case)
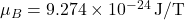 ,
, 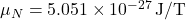 , proton moment
, proton moment 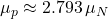 .
.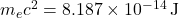 ,
, 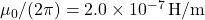 .
.
![]()
![]()